Welcome to Phazer Inc.
Here at Phazer Inc., we aim to use our groundbreaking technologies to explore the science behind the phases of matter. It's not rocket science. Oh wait, it is. We test theories surrounding philosophy, lemonology, and archery to bring ideas into reality. We believe that by understanding the process of phase changes and researching solid, liquid, gas and plasma, we can achieve greatness.

Phase Changes
Plasma
- Deionization - A phase change where charged particles (electrons & ions) combine into a neutral gas state.
Solid
- Sublimation - A phase change where a solid transitions directly to a gas without the typical melting and boiling sequence.
- Melting - A transition from solid to liquid once at the melting point.
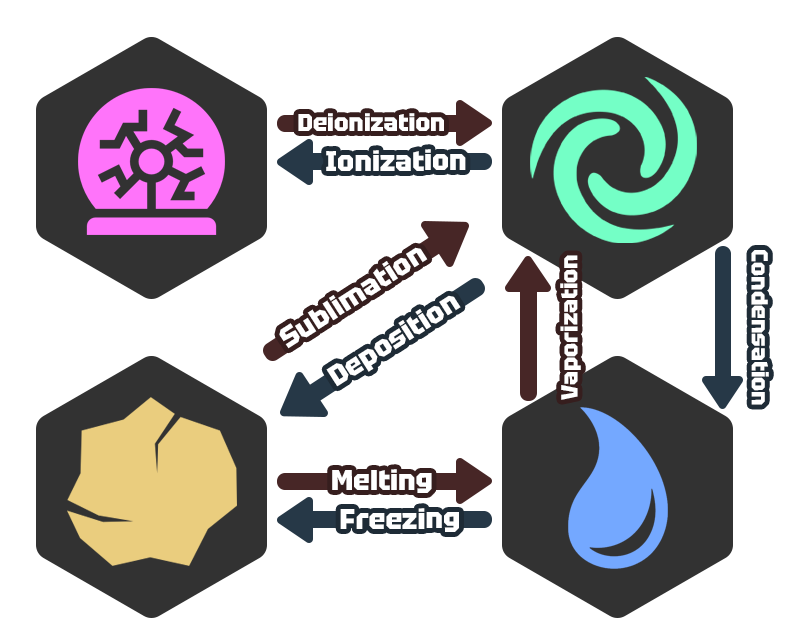
Gas
- Ionization - A transition from gas to plasma through added energy, stripping electrons from atoms.
- Deposition - An immediate transition from gas to solid when gas particles lose energy.
- Condensation - A phase change where gas loses energy (typically by cooling down) and forming liquid droplets.
Liquid
- Vaporization - An endothermic process where liquid gains enough kinetic energy to escape into the air.
- Freezing - An exothermic transition from liquid to solid when the temperature falls below the substance's freezing point.
Solid
A solid is a substance where the molecules are closely bound together due to molecular forces. Solids hold their shape and have fixed volumes, with rigid particles that are locked into place. Particles in these substances are not easily compressible and have little free space.

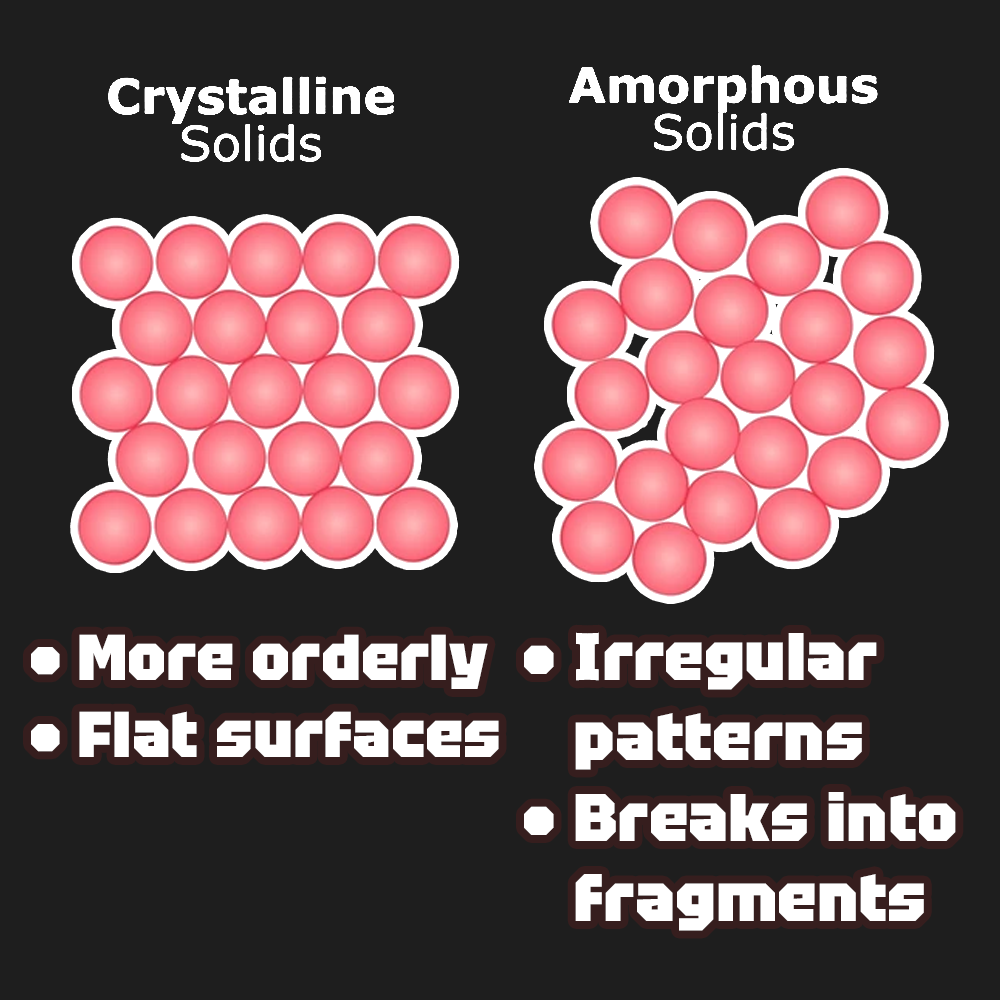
Two types of solids include crystalline solids and amorphous solids. Crystalline solids typically have highly ordered repeating structures, leading to distinct flat surfaces or faces. Amorphous solids produce fragments when cleaved or broken. They have poorly defined patterns, since their components are not arranged in a regular array.
Liquid
A liquid is a substance with particles that can move or slide past each other. Typically, they assume the shape of the container they are put in, meaning they have a definite volume without a fixed form. Liquids do not easily compress and can flow easily.


There are several different forms of liquids with different molecular interaction and flow behavior. Newtonian liquids have a constant viscosity, non-Newtonian liquids have a viscosity that changes with force, and superfluids are fluids with zero viscosity (usually seen near absolute zero) that prevent the loss of kinetic energy.
Gas
A gas is a substance with particles that move and vibrate at high speeds. These particles are highly separated, having an unordered arrangement. Gas, similar to liquid, is able flow easily without needing to maintain a fixed shape.


There are various types of gases that differ by composition and reactivity. Some notable types include noble gases, which are unreactive, monatomic elements, elemental gases comprised of a single element (e.g. Hydrogen, Nitrogen), and compound gases, which are molecules composed to two or more elements (e.g. Carbon Dioxide, Carbon Monoxide).
Plasma
Plasma is a superheated form of gas where electrons are freed from their atoms, allowing the substance to react to electric and magnetic fields. Some examples of plasma include lightning, stars such as the sun, auroras and fusion reactors.

Fusion is the power used by the sun and stars, which combines light elements in the form of plasma. Scientists look to utilize fusion in order to create a seemingly infinite supply of energy. Currently, researchers aim to build fusion reactors that maximize the amount of ions in a small region and the duration that they stay close together. To do this, they heat plasmas to temperatures even hotter than the core of the sun.






